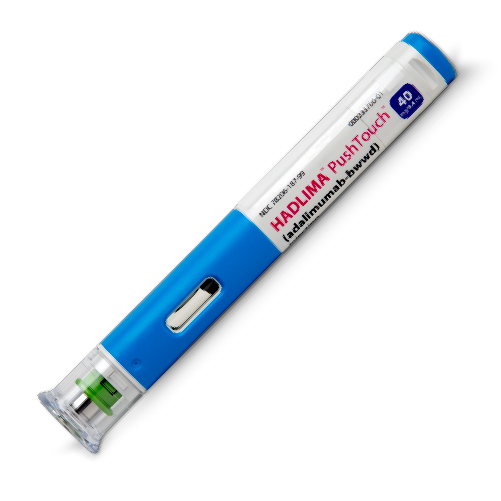
HADLIMA™ (adalimumab-bwwd): PushTouch™ Autoinjector
Designed with the patient in mind, this single-use, sure-grip autoinjector is intended to fit into a patient’s lifestyle.
The HADLIMA™ PushTouch™ Autoinjector is a single-use device for administering HADLIMA, an FDA-approved biosimilar to HUMIRA (adalimumab).
The HADLIMA™ PushTouch™ Autoinjector was specifically designed with the patient in mind, with a thin 29-gauge needle, a latex-free needle cover, and a buttonless device with a sure-grip shape and non-slipsurface that is intended to fit into a patient’s lifestyle.